Sales of $4.2 million and Profitability
MISSISSAUGA, May 11, 2023 – Microbix Biosystems Inc. (TSX: MBX, OTCQX: MBXBF, Microbix®), a life sciences innovator, manufacturer, and exporter, reports results for its second quarter and first half of fiscal 2023 ended March 31, 2023 (“Q2” and “H1”), a period in which a loss was incurred in the first quarter and profitability restored in Q2. Microbix remains well-capitalized and is targeting to continue increasing sales of its testing-related medical devices and ingredients, thereby growing sales, margins, and earnings.
Management Discussion
Q2 sales of $4.2 million reflected a recovery in sales of test ingredients, flattish sales of test-controls (“QAPs”) and none of sample collection media (“DxTM™”). The resulting gross margin dollars and control of expenditures enabled a small profit for Q2. For the balance of fiscal 2023, Microbix expects increased sales of both Antigens and QAPs to enable it to generate positive net earnings for the full-year – for a third consecutive year of profitability while further growth-oriented initiatives continue to be executed.
For the three months ending March 31, 2023 (“Q2”)
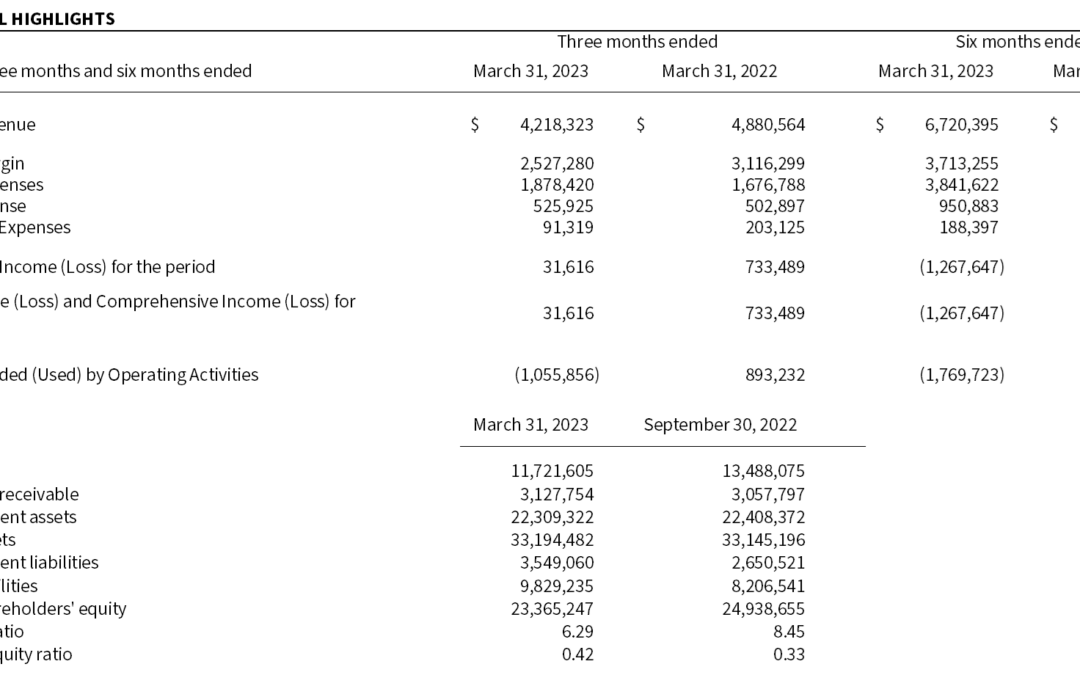
Q2 revenue was $4,218,323, a decrease from Q2 2022 revenues of $4,880,564. Included were antigen revenues of $3,004,730 (2022 – $1,607,970). QAPs revenues were $1,101,684 (2022 – $1,318,382). In turn, revenue from DxTM was zero in Q2 (2022 – $1,860,704), and royalties were $111,910 (2022 – $93,508). The Q2 2022 sales mix and total was most greatly influenced by the recovery of antigen sales and a lack of Ontario-driven deliveries of DxTM.
Q2 gross margin was 60%, down slightly from 64% in Q2 2022 due to a greater proportion of lower margin antigen test-ingredient sales, which tend to have lower margins than fully-regulated and branded medical devices such as QAPs and DxTM. In addition, we continue to see double digit increases in our supply chain costs, which can only be passed-through to end-customers over time.
Q2 Operating expenses increased by 5% relative to 2022, principally due to investment in IT infrastructure to support growth objectives – namely start-up costs relating to our “ERP” and “eQMS” implementations. Finance expenses were lower due to repayment of debentures and long-term debt during 2022 and short-term investment of cash balances. Overall, Q2 sales led to an operating profit and net income of $31,616 versus a Q2 2022 operating income and net income of $733,489. Cash used in operating activities was $1,055,856, compared to cash provided by operating activities of $893,232 in Q2 2022, with the majority of the change coming from deployment of cash into working capital accounts during the quarter.
Six Months Ending March 31, 2023 (“H1”)
H1 revenue was $6,720,395, a 31% decrease from H1 2021 revenues of $9,736,164. Included were antigen revenues of $4,008,537 (2022 – $3,374,386), up 19% from last year. QAPs revenues of $2,435,186 were flat year-over-year (2022 – $2,467,533) due largely to delays in test finalization and launch timelines of customers intending to incorporate Microbix’s QAPs in their kits of test consumables. In turn, revenue from DxTM was zero (2022 – $3,677,949), and royalties were $276,672 (2022 – $216,296). As a whole, the lower H1 sales result was most driven by the lack of deliveries of DxTM for the Province of Ontario.
H1 gross margin was 55%, down from 65% in H1 2022, due to the lack of VTM sales and the effects of a greater proportion of antigen sales that have lower margins. In addition, we continue to see double digit materials price increases across our supply chain.
Operating expenses in H1 increased by 5% relative to H1 2022, due to increased investment in IT infrastructure that was partly offset by lower finance expenses due to repayment of debentures and long-term debt during fiscal 2022 and short-term investment of cash balances. Overall, weaker H1 sales led to an operating loss and net loss of $1,267,647 versus a H1 2022 operating income and net income of $1,614,267. Cash used in operating activities was $1,769,723, compared to cash provided by operating activities of $609,218 in H1 2022, with much of the change coming from changes in working capital balances during the period.
At the end of H1, Microbix’s current ratio (current assets divided by current liabilities) was 6.29 and its debt to equity ratio (total debt over shareholders’ equity) was 0.42.
Corporate Outlook
Microbix will continue working to drive sales growth across its three revenue-generating business lines, and on improving percentage gross margins and driving bottom-line results. Management currently expects Microbix to generate meaningful net earnings across full-year fiscal 2023. Additionally, work continues upon securing a partnership to advance its Kinlytic® urokinase project.
Adelaide Capital will host a live webinar with management on Tuesday, May 16 at 11am ET. Please register here: https://us02web.zoom.us/webinar/register/WN_VCJbOVd8RAufxG0tdhqGIw. It will also be live-streamed to YouTube at: https://www.youtube.com/channel/UC7Jpt_DWjF1qSCzfKlpLMWw.
A replay of the webinar will also be made available on Adelaide Capital’s YouTube channel.
About Microbix Biosystems
Microbix creates proprietary biological products for human health, with over 100 skilled employees and annualized sales targeting C$ 2.0 million per month. It makes a wide range of critical ingredients and devices for the global diagnostics industry, notably antigens for immunoassays and its laboratory quality assessment products (QAPs™) that support clinical lab proficiency testing, enable assay development and validation, or help ensure the quality of clinical diagnostic workflows. Its antigens drive the antibody tests of over 100 diagnostics makers, while QAPs are sold to clinical laboratory accreditation organizations, diagnostics companies, and clinical laboratories. Microbix QAPs are now available in over 30 countries, distributed by 1WA (Oneworld Accuracy Inc.), Alpha-Tec Systems, Inc., Diagnostic International Distribution SpA., Labquality Oy, The Medical Supply Company of Ireland, Neo-Science Equipment LLC, R-Biopharm AG, SDT Molecular Pte Ltd, Seegene Canada Inc., and Thomas Scientific LLC. Microbix is ISO 9001 and 13485 accredited, U.S. FDA registered, Australian TGA registered, Health Canada establishment licensed, and provides CE marked products.
Microbix also applies its biological expertise and infrastructure to develop other proprietary products and technologies, most notably viral transport medium (DxTM™) to stabilize patient samples for lab-based molecular diagnostic testing and Kinlytic® urokinase, a biologic thrombolytic drug used to treat blood clots. Microbix is traded on the TSX and OTCQX, and headquartered in Mississauga, Ontario, Canada.
Forward-Looking Information
This news release includes “forward-looking information,” as such term is defined in applicable securities laws. Forward-looking information includes, without limitation, discussion of financial results or the outlook for the business, risks associated with its financial results and stability, its current or future products, development projects such as those referenced herein, sales to foreign jurisdictions, engineering and construction, production (including control over costs, quality, quantity and timeliness of delivery), foreign currency and exchange rates, maintaining adequate working capital and raising further capital on acceptable terms or at all, and other similar statements concerning anticipated future events, conditions or results that are not historical facts. These statements reflect management’s current estimates, beliefs, intentions and expectations; they are not guarantees of future performance. The Company cautions that all forward looking information is inherently uncertain and that actual performance may be affected by a number of material factors, many of which are beyond the Company’s control. Accordingly, actual future events, conditions and results may differ materially from the estimates, beliefs, intentions and expectations expressed or implied in the forward-looking information. All statements are made as of the date of this news release and represent the Company’s judgement as of the date of this new release, and the Company is under no obligation to update or alter any forward-looking information.
Please visit www.microbix.com or www.sedar.com for recent Microbix news and filings.
For further information, please contact Microbix at:
| Cameron Groome, CEO
(905) 361-8910 |
Jim Currie,
CFO (905) 361-8910 |
Deborah Honig,
Investor Relations Adelaide Capital Markets (647) 203-8793 ir@microbix.com |
Copyright © 2023 Microbix Biosystems Inc. Microbix®, DxTM™, Kinlytic®, and QAPs™ are trademarks of Microbix Biosystems Inc.